SPG graphene special spray engineering
Graphene is a two-dimensional crystal with a thickness of one atom, which is separated from the graphite material and composed of carbon atoms. Graphene narrowly refers to a single layer of graphite with a thickness of 0.335 nm and only one layer of carbon atoms. In practice, however, a graphite structure within 10 layers may also be referred to as graphene, and a layer of 10 or more is referred to as a graphite film. Single-layer graphene refers to graphite having a thickness of only one carbon atom layer, and carbon atoms-carbon atoms are connected by covalent bonds to form a honeycomb structure. The perfect graphene has an ideal two-dimensional crystal structure consisting of a hexagonal lattice with a theoretical specific surface area of up to 2.6 × 102 m2 / g. Graphene has excellent thermal conductivity (3×103 W/(m•K)) and mechanical properties (1.06×103 GPa). In addition, the graphene-stabilized regular hexagonal lattice structure gives it excellent electrical conductivity, and the electron mobility at room temperature is as high as 1.5 × 104 cm 2 / (V • s). The special structure of graphene, outstanding thermal conductivity and mechanical properties have become a hotspot in materials science research.

Graphene structure
Graphene refers to a honeycomb crystal structure in which only one atomic-scale thick single-layer graphite layer is closely arranged by sp2 hybridized carbon atoms. The carbon-carbon bond length in graphene is about 0.142 nm. There are three sigma bonds in each crystal lattice, and the connection is very strong to form a stable hexagon. The π bond perpendicular to the direction of the crystal plane plays a large role in the process of graphene conduction. Graphene is the basic building block of graphite, carbon nanotubes and fullerenes. It can be regarded as an infinite aromatic molecule. The limit of planar polycyclic hydrocarbons is graphene.
In terms of image, graphene is closely packed into a two-dimensional honeycomb lattice structure by a single layer of carbon atoms, which looks like a plane formed by a hexagonal grid. In a single-layer graphene, each carbon atom is rectified by a sp2 hybridization bond with a surrounding carbon atom. Each hexagonal unit is actually similar to a benzene ring, and the carbon atoms contribute an unbonded electron. The thickness of the single-layer graphene is only 0.35 nm, which is about one-hundred thousandth of the diameter of the hair.
The structure of graphene is very stable, and the connection between carbon atoms is flexible. When subjected to external forces, the carbon atom plane will be bent and deformed, so that the carbon atoms do not have to be rearranged to adapt to external forces, thereby ensuring their structural stability.
Graphene is a finite structure and can exist in the form of nanoscale strips. When the charge moves laterally in the nanoribbon, an energy barrier is generated near the neutral point, and the barrier increases as the strip width decreases. Therefore, the desired barrier can be further obtained by controlling the width of the graphene strip. This feature is the basis for the development of graphene-based electronic devices.
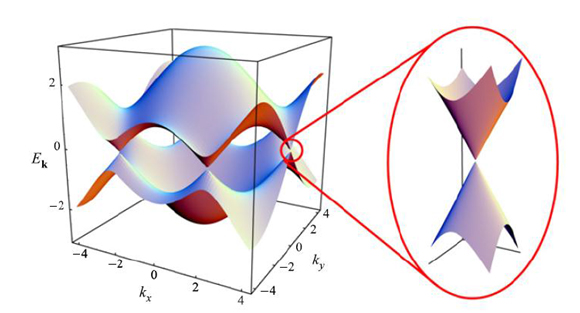
Graphene performance
Graphene is an ultra-light material with an areal density of 0.77 mg/m2. The main properties are: First, it has excellent electrical conductivity. Graphene has a higher electron mobility than carbon nanotubes or silicon crystals, 100 times that of silicon, and can reach 15 000 cm 2 /( V•s) at room temperature. The resistivity is much lower than aluminum, copper and silver, and is only about 10 to 6 Ω•cm. Second, it has super thermal conductivity. The thermal conductivity of graphene is better than that of carbon nanotubes, which is 10 times that of metals such as copper and aluminum, and the thermal conductivity is as high as 5300 W/m•K. Third, it has superior mechanical properties. The hardness of graphene exceeds that of diamond, and the breaking strength reaches 100 times that of steel. Fourth, it has super light transmission. The light absorption of the graphene is very small, and the light transmittance is as high as 97.7%. The fifth is to have a strong specific surface area. The specific surface area of graphene is 1130 m2 per gram higher than that of ordinary activated carbon, reaching 2630 m 2 /g.
Optical properties of graphene
Graphene is known to be the thinnest and hardest nanomaterial in the world. It is almost completely transparent, absorbing only 2.3% of light and has excellent optical properties. Theoretical and experimental results show that single-layer graphene absorbs 2.3% of visible light, that is, the transmittance is 97.7%. The visible light transmittance from the substrate to the single-layer graphene and the double-layer graphene differs by 2.3% in order, so that the number of layers can be estimated from the visible light transmittance of the graphene film. Combined with the non-interactive Dirac-Feomizi theory, simulating the transmittance of graphene, the results consistent with the experimental data can be obtained.
According to the principle of refraction and interference, graphene of different layers will show different colors and contrasts under the optical microscope, which is convenient for distinguishing the number of graphene layers.
Theoretical and experimental results show that the large-area graphene film also has excellent optical properties, and its optical properties vary in the thickness of the graphene. Graphene film is a kind of typical transparent conductive film, which can replace traditional thin film materials such as indium tin oxide (ITO) and fluorine-doped indium oxide (FTO), which can overcome the brittle defects of ITO film and solve the scarcity of indium resources. Limitations and many other issues. The graphene transparent conductive film can be used as a window layer electrode of a dye-sensitized solar cell and a liquid crystal device.
In addition, when the intensity of incident light exceeds a certain critical value, the absorption of graphene will be saturated. This nonlinear optical behavior becomes saturated absorption. In the near-infrared region, under strong light irradiation, graphene will slowly approach saturation absorption due to its broad band absorption and zero band gap characteristics. Using this property, graphene can be used for ultra-fast photonics, such as fiber lasers.
Electrical properties of graphene
Each carbon atom of graphene is sp2 hybridized, and contributes to the remaining one p-orbital electron to form a π bond, and the π electron can move freely, giving the graphene excellent conductivity. Since the interaction between atoms is very strong, at normal temperature, even if the surrounding carbon atoms collide, the electrons in the graphene receive little interference. When electrons are transported in graphene, scattering is less likely to occur, and the transmission efficiency is 1.5×105 cm 2 /( V•s), which is about 140 times that of electron mobility in silicon. It has a conductivity of up to 106 s/m and a resistivity of only about 10-6 Ω•cm, which is lower than copper or silver and is the world's smallest resistivity material. Because of its extremely low resistivity, electron migration is extremely fast, so it is expected to be used to develop a new generation of electronic components or transistors that are thinner and faster. Since graphene is essentially a transparent, good conductor, it is also suitable for making transparent touch screens, light panels, and even solar cells.
The emergence of graphene has ignited a huge wave in the scientific community. It has been found that graphene has extraordinary electrical conductivity, more than ten times the strength of steel and excellent light transmission, and its emergence is expected to trigger a revolution in the field of modern electronic technology. In graphene, electrons can migrate very efficiently, while conventional semiconductors and conductors, such as silicon and copper, perform far better than graphene. Due to the collision of electrons and atoms, traditional semiconductors and conductors release some energy in the form of heat. In 2013, general computer chips waste 72%-81% of electricity in this way. Graphene is different, and its electron energy is not. It will be worn out, which gives it an unusually good character.
Mechanical properties of graphene
Graphene is the crystal structure with the highest strength and hardness in the material. Its tensile strength and modulus of elasticity are 125 GPa and 1.1 TPa, respectively. The strength limit of graphene is 42 N/m2. The ideal graphene has a strength of about 100 times that of ordinary steel, and the graphene layer with an area of 1 m2 can withstand a mass of 4 kg. Graphene can be used as a typical two-dimensional reinforcement material and has potential application value in the field of composite materials.
Thermal properties of graphene
Graphene is harder than diamond and retains its original shape at high temperatures. This shocks the physical world, mainly because the carbon atoms in the graphene are regularly and regularly applied. When an external force acts on the graphene, the internal carbon atoms do not shift, but only when the bending deformation occurs, the external force can be resisted to ensure the stability.
The room temperature thermal conductivity of graphene is more than 10 times that of copper at room temperature, and the thermal conductivity is 5300 W/m•K, which is higher than that of carbon nanotubes and diamond. The theoretical specific surface area of graphene can reach 2630m2/g. The micro-sensor supported by graphene can sense a single atom or molecule. When the gas adheres or leaves the surface of graphene, the adsorbed molecules change the local carrier concentration of graphene. This causes a step change in the resistance. This feature can be used to make gas sensors. Theoretical calculations show that graphene and lithium can form a porous composite structure and have a strong hydrogen storage capacity.
Magnetic properties of graphene
Graphene tends to have ferromagnetism after hydrogenation, mainly because graphene has a pair of electron pairs at the edges after hydrogenation, which makes graphene magnetic. The researchers also did by changing the temperature in the presence of a magnetic field to see if the magnetic properties of graphene could be changed. The magnetic field strength is determined to be 1T. When the temperature is T<90K, the graphene exhibits a paramagnetic characteristic; when the temperature T>90K, the graphene exhibits a diamagnetic characteristic.
Chemical properties of graphene
The electronic properties of graphene have received extensive attention. However, the chemical properties of graphene have been left untouched. To date, we only know about the chemical properties of graphene: graphene can adsorb atoms and molecules around (eg : Nitrogen dioxide, ammonia, potassium), this property is somewhat similar to the activated carbon we know. Nitrogen dioxide, ammonia, and potassium are often used as donors or acceptors to change the concentration of carbon atoms inside the graphene. However, graphene itself is a conductive material. Other adsorbates, such as hydrogen and hydroxide ions, produce poorly conductive derivatives, but these are not new compounds, but graphenes decorate different adsorbates. Since graphene and graphite are both allotropes of carbon, from the chemical point of view, they often have some of the same properties, so in some areas where graphene is unfamiliar, graphite can be used to carry out corresponding experiments to find graphite. The law of olefins, with this simple and convenient idea, in the future, more chemical properties of graphene will be discovered.
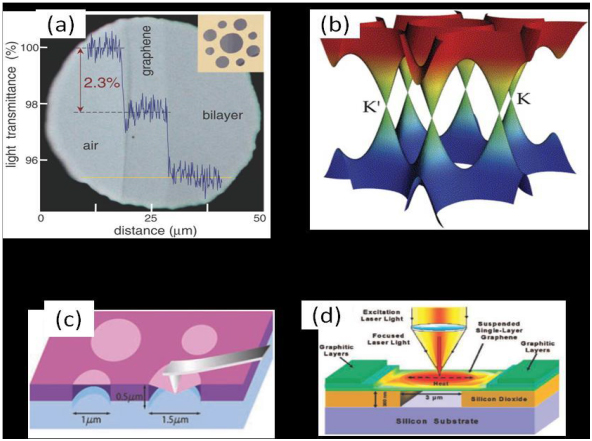
Schematic diagram of optical, electrical, mechanical and thermal properties of graphene
Our company independently develops and manufactures graphene special dryer according to the performance characteristics of graphene. Welcome to inquire for detailed technology!
- 2018-12-05 > Comparison of powder extraction rate between traditional Chinese medicine extract and high speed spray dryer
- 2018-10-30 > Safe use of hot air circulation oven and four points of attention
- 2018-09-10 > The rice dryer market will also see new trends
- 2018-09-06 > Analysis on the Application of Eight Drying Equipments in Chemical Pharmaceutical Industry
- 2018-05-24 > Application of spray drying desulfurization technology in protecting ecological environment
- 2018-02-20 > Belt dryers are common faults and maintenance during use
- 2017-12-10 > Application range and selection of belt dryer
- 2017-08-22 > Development status and characteristics of high efficiency mixing granulator for calcium carbonate industry
- 2017-03-29 > Four major process design methods for rotary flash dryer














